Simultaneous Global New Drug Development: Multi-Regional Clinical Trials after ICH E17
Gang Li, Bruce Binkowitz, William Wang, Hui Quan, Josh Chen
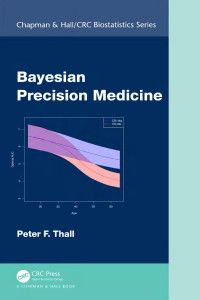
This book contains chapters providing interpretations of principles in ICH E17 and new ideas of implementing MRCTs. Authors are from different regions, and from academia and industry. This book will be of particular interest to biostatisticians working in late stage clinical development of medical products.
Jahr:
2021
Auflage:
1
Verlag:
Chapman & Hall/CRC
Sprache:
english
Seiten:
304
ISBN 10:
1000485064
ISBN 13:
9781000485066
Serien:
Chapman & Hall/CRC Biostatistics Series
Datei:
PDF, 14.82 MB
IPFS:
,
english, 2021
 Amazon
Amazon  Barnes & Noble
Barnes & Noble  Bookshop.org
Bookshop.org  Konvertieren Sie Dateien
Konvertieren Sie Dateien Mehr Suchergebnisse
Mehr Suchergebnisse Andere Vorteile
Andere Vorteile